Bacteriophage Therapy
Nature’s Precision Weapon Against Infection
This is a rich topic that sits at the intersection of microbiology, drug resistance, regulatory policy, and national health strategy. Let me pull it together into a coherent narrative.
Despite a career in computers, not biology, I spent the last ten years of my career as a computer architect for a major biotech company, where I found that the vertical specialisation that I had fought in the computer industry (Windows, MacOS, Linux, C, JAVA, Python, etc) was a normal resistance to change. Once you had reached competence in one subject, you were resistant to new ideas.
As an architect, I was a problem solver, not a disciple of any solution. So, as I learned about antibiotics and the problems with their use, as they mutated and became less effective over time, I decided that my computer designs had to be flexible enough to be used if another biological technology were to be used.
After some research, I discovered an alternative to antibiotics that has been available for over 100 years. Bacteriophages or as they are normally called, Phages.
The Antimicrobial Resistance (AMR) Crisis That Drove the Renaissance
The context for the renewed interest in phage therapy is stark. Over the next 25 years, an estimated 39 million people are expected to die from drug-resistant pathogens alone. The United Nations General Assembly, convening in 2016, deemed antimicrobial resistance “the greatest and most urgent global risk.” Against that backdrop, medicine has been compelled to revisit a therapeutic tradition it largely abandoned seventy years ago.
What a Bacteriophage Actually Is
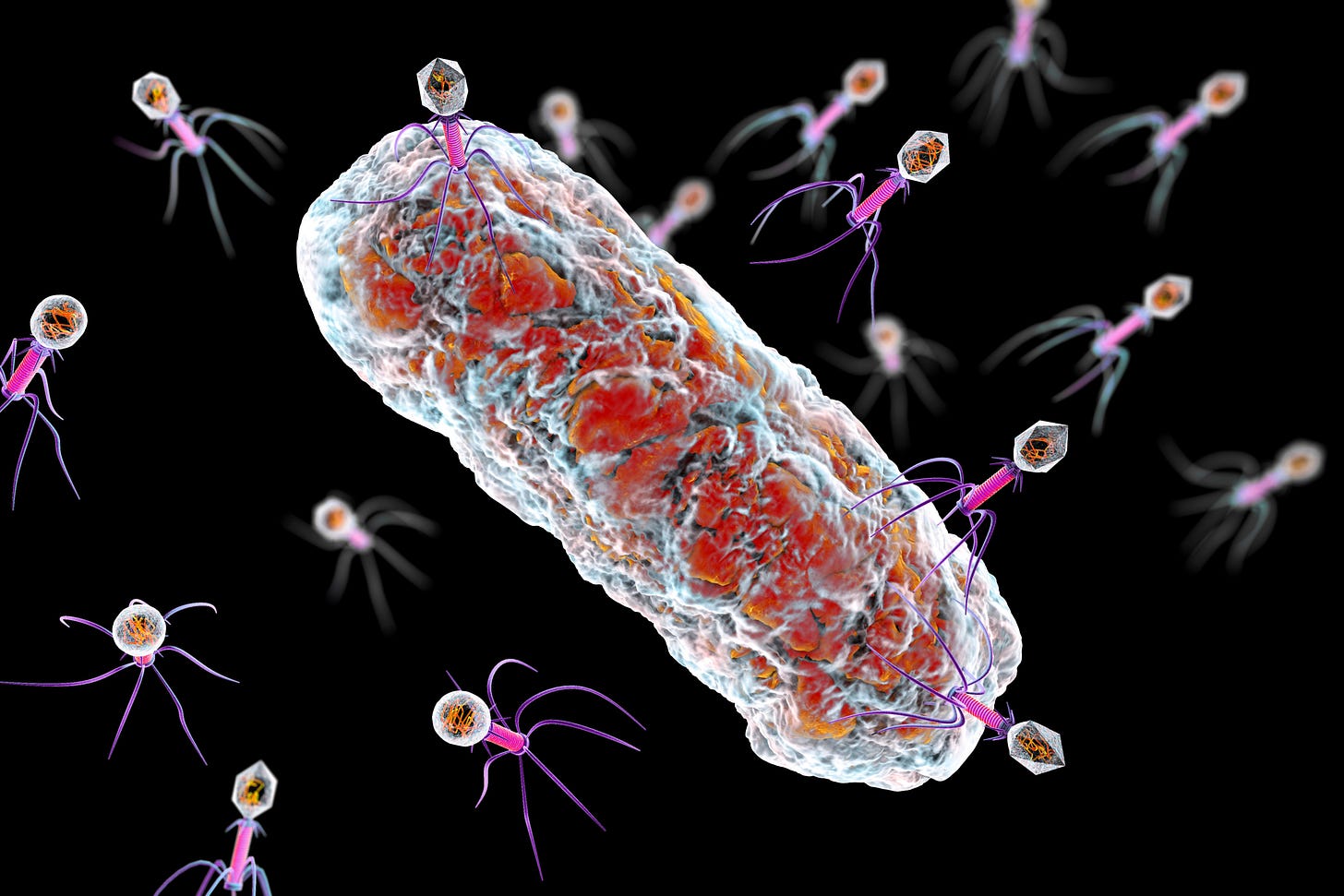
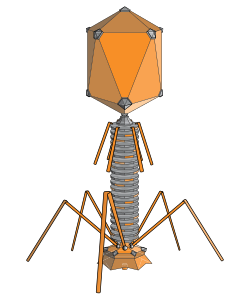
A bacteriophage — “phage” for short — is a virus that infects bacteria rather than human cells. Phages attach to bacterial cells and inject their genome into the cell. The bacteria’s production of the viral genome interferes with its ability to function, halting the bacterial infection. The bacterial cell causing the infection is unable to reproduce and instead produces additional phages. In effect, the phage hijacks the bacterium’s own machinery to replicate, then bursts it open in what is called the lytic cycle. Approximately 10³¹ bacteriophages are present in the world, outnumbering bacteria by an estimated tenfold.
A History Interrupted by Penicillin
Bacteriophages were discovered by two scientists working independently: Frederick W. Twort in Great Britain in 1915, and Canadian Félix d’Hérelle in France in 1917. Both immediately recognised the therapeutic potential. In 1919, the first documented use of bacteriophages to treat bacterial infections was reported, and thereafter, numerous reports described their successful use in clinical and pre-clinical settings.
Phage therapy was progressively abandoned since the late 1930s following the rise of antibiotics, which were much easier and cheaper to produce and use. Western medicine essentially dropped the entire field. The Soviet Union and its satellite states did not, however. The Eliava Institute in Georgia continued phage therapy throughout the Cold War and successfully treated patients with skin ulcers, infected burn wounds, stomach bugs, eye infections, and respiratory infections. Georgia remains one of the few places today where phage therapy is genuinely embedded in medical practice.
Phage therapy has largely existed on the fringes of medicine, particularly in Western countries, where it is occasionally approved for compassionate use — that is, used on an emergency basis when no other approved therapies are available. The most prominent such case was that of Tom Patterson, a professor at UC San Diego, who in 2015 contracted a deadly multidrug-resistant Acinetobacter baumannii infection. No antibiotics could control his infection. The team relied on researchers from the Center for Phage Technology at Texas A&M University, as well as scientists from the US Navy, to find phages that could kill his bacterial isolate. His recovery made international headlines and arguably did more than any published paper to reawaken Western medical interest.
The Advantages Over Conventional Antibiotics
The case for phages rests on several overlapping properties, each of which addresses a specific weakness of antibiotic therapy.
Precision targeting. Each phage has evolved to narrowly target bacterial strains or species. In contrast to many antibiotics, which obliterate harmful bacteria while simultaneously decimating the microbiota (thus triggering a new set of problems), phage therapy is an attractive alternative for managing infections, especially those caused by multidrug-resistant bacteria. Broad-spectrum antibiotics are the medical equivalent of a carpet bomb; phages are a guided munition.
Self-amplification. During the bacterial-killing process, phages can increase in number specifically where hosts are located. This self-amplification means that the phage dose effectively grows where it is most needed. This is pharmacologically without parallel — the drug increases its own concentration precisely at the site of infection.
Biofilm penetration. Biofilm infections are very challenging to treat with antibiotics. The biofilm matrix and surrounding bacterial membranes can bind to the antibiotics, preventing them from penetrating. The matrix may contain enzymes that deactivate antibiotics, and biofilms also have low metabolic activity. Phages, by contrast, have a demonstrated ability to clear at least some biofilms, perhaps owing to their potential to actively penetrate by lysing one bacterial layer at a time, or due to the display of biofilm exopolymer-degrading depolymerases. This is critical for the treatment of infections associated with implanted medical devices, diabetic foot ulcers, and chronic lung infections in cystic fibrosis.
Resistance dynamics. Bacteria can and do evolve resistance to phages, but the dynamics differ from those of antibiotic resistance. Viruses can also evolve, meaning they can potentially produce more varieties of phages capable of killing even superbugs. This co-evolutionary arms race can, under the right conditions, be exploited therapeutically — resistance to phages can paradoxically make some bacteria less virulent or more susceptible to antibiotics.
Phage-antibiotic synergy. Phages and antibiotics need not be competitors. Phage-antibiotic synergy is the phenomenon in which the combined effect is greater than the sum of the effects of either alone. Phage therapy can sensitise bacteria to antibiotics, lowering the minimum inhibitory concentration and increasing susceptibility, while phages can also degrade bacterial polysaccharides, aiding antibiotic diffusion.
Low inherent toxicity. Because phages target bacteria so specifically, they have low toxicity to humans. The human body already carries enormous numbers of phages — estimates suggest trillions in and on us at any given moment — meaning the immune system has extensive experience coexisting with them.
The Technical Challenges
Despite all these advantages, phage therapy faces genuine and substantial obstacles. These are not merely regulatory foot-dragging; many are intrinsic to the biology.
Narrow host range. The same precision that makes phages attractive also constrains them. Usually, a specific bacteriophage infects and kills only one bacterial species. This means a clinician must first identify the causative organism with considerable accuracy, then match a phage to it. The major disadvantage of phage therapy is the need to rapidly and accurately determine the precise etiological microorganism causing the infection. A clinical sample must be isolated and cultured using standard microbiology diagnostic procedures to identify the pathogen before a specific bacteriophage solution can be defined and later administered to the patient. In a septic patient, that time pressure can be life-threatening.
Phage cocktails and the matching problem. Phage mixtures (”cocktails”) are sometimes used to improve the chances of success. But designing an effective cocktail and knowing which phages within it are doing the work is complex. AI is increasingly being brought to bear on this problem: researchers at the Institut Pasteur developed and trained an AI model capable of making a tailored selection of phages based solely on the genome of the targeted bacteria, achieving a 90% success rate in destroying selected E. coli strains in testing.
Temperate phages and transduction. Not all phages are killers in the simple sense. In the lysogenic cycle, so-called temperate phages insert their genetic material into the bacterial chromosome, where it remains silent for extended periods and is replicated along with the chromosome. More concerning, some phages can transfer bacterial DNA from one host to another through generalised transduction. With some well-characterised phages, up to 5% of the virus particles contain only bacterial DNA, meaning the entire genome of the propagating host is present in more than a million copies per millilitre of a typical preparation. Only rigorously characterised lytic phages are safe for therapeutic use.
Immune response. The body can mount an immune response against phages, potentially neutralising them before they reach their target, especially on repeated administration. The relationship between phages and the immune system is complex: phage therapy can also act as an anti-inflammatory agent to control cytokine storms, but this dual immunological role means effects are difficult to predict.
Manufacturing and clinical trials. Phage therapies may be too personalised to easily allow large-scale clinical trials. If a phage treatment involves a mix of phage types and other drugs specific to the patient and infection, it becomes hard to run double-blind, placebo-controlled trials. The conventional pharmaceutical development pipeline was simply not designed for medicines that are alive, evolving, and tailored to individuals.
Cost. Phages can be contract-manufactured according to Good Manufacturing Practice (GMP), but at a cost of over £500,000 for phages sufficient for a few hundred patients. Until scalable, sustainable manufacturing is established, the economics are prohibitive.
The UK Position: Promising Infrastructure, Patchy Funding
The UK has a historically notable claim on phage research — Twort’s original 1915 discovery was made in Britain — but it has been slow to translate contemporary interest into clinical infrastructure. The picture is improving, though unevenly.
Institutional landscape. The most significant academic centre is the Leicester Centre for Phage Research (LCPR) at the University of Leicester, which has built one of the UK’s largest and best-characterised phage collections. The LCPR and the University of Northumbria have established a distributed phage bank between the two universities, with expansion plans contingent on additional funding. A new organisation, UK Phage Therapy, is working in partnership with the LCPR, the Centre for Process Innovation (CPI), and Fixed Phage Limited to establish a national clinical and manufacturing infrastructure. In the last two years, 12 patients in the UK have received phage therapy, and there is burgeoning clinical interest. All cases currently depend on importing phages from abroad — a situation that is clearly unsustainable.
Parliamentary scrutiny and government response. The UK Science, Innovation and Technology Committee undertook a formal inquiry into the antimicrobial potential of bacteriophages, and the government published its response in March 2024. The tone was supportive but non-committal. The government is committed to exploring alternative therapies, including bacteriophages, to tackle antimicrobial resistance, but is reluctant to prioritise any single technology for funding or reporting. Phages have been included in the 2024–2029 AMR National Action Plan, but multiple hurdles still hinder the implementation of phage therapy in mainstream national medicine, and currently, no phages are licensed in the UK.
Regulatory breakthrough. A significant moment came in June 2025, when the MHRA published the UK’s first official guidance to support the safe development and use of phage therapies. The guidance covers both combined phage products designed for common infections and personalised phage therapies tailored for individual patients with rare or highly resistant infections. This had been a major bottleneck: developers needed to know what regulatory pathway to follow before committing investment.
Public funding. A total of approximately £67.5 million of public funding has been awarded across 144 projects focused on phage research and development since 2006, from investigations into basic phage biology through to the development of phage-based products and scaling manufacturing processes. NIHR does not routinely ringfence funding for specific conditions or research areas but welcomes funding applications for research into any aspect of human health, including bacteriophages, with awards made on the basis of importance to patients, value for money, and scientific quality.
Private sector hesitation. Pharmaceutical companies are unwilling to invest, in part because patenting phages — which exist in nature — can be tricky, although genetically engineered phages might be easier to patent. This is a structural problem. The business model that drives antibiotic development — broad-spectrum products scalable to millions of patients — does not map onto highly personalised phage preparations.
Manufacturing is the central bottleneck. The critical missing piece is domestic GMP manufacturing capacity. The provision of phage therapy in the UK will not progress beyond an increasing number of ad hoc cases until an onshore sustainable and scalable source of GMP phages is established. In countries where phage therapy has progressed clinically and commercially, this has typically been underpinned in some form by public funding. A transformative £520 million fund over five years from 2025 to 2030 has been announced to support life sciences manufacturing investment in the UK more broadly, and the government has committed to considering whether a dedicated GMP facility specifically for phage innovators could be supported.
November 2025 conference. In a sign of genuine momentum, on 25 November 2025 the international phage therapy research and biotechnology communities gathered in Liverpool to discuss progress in implementing clinical phage therapies and the latest technological breakthroughs in phage biology and engineering, while UK regulators and health agencies provided the latest guidance.
Conclusion
Phage therapy represents one of those rare cases where medicine has inadvertently discarded a powerful tool, and is now rediscovering it under rather urgent circumstances. The science is compelling: precision targeting, self-amplifying dosing, biofilm penetration, and co-evolutionary adaptability are properties no antibiotic can claim. The obstacles are real but not insurmountable — they are principally obstacles of manufacturing scale, regulatory novelty, and the mismatch between personalised medicine and standardised clinical trial design.
The UK is better positioned than most countries to lead this field — it has strong academic research, the MHRA’s new guidance framework, a nascent national phage library, and a political class that now at least acknowledges the issue. What it lacks, as yet, is the sustained public investment and dedicated manufacturing infrastructure that would move the field from compassionate use edge cases to a genuine part of the NHS therapeutic toolkit. Given that AMR is projected to be one of the defining medical crises of the mid-21st century, the opportunity cost of continued hesitation is rising.